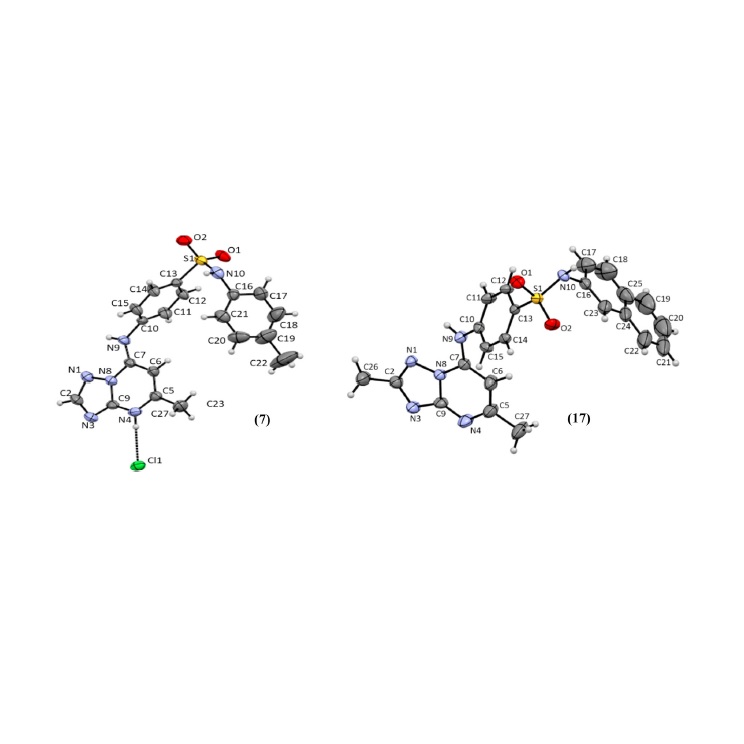
 Abstract: In this work, we designed and synthesized 35 new triazolopyrimidine, pyrazolopyrimidine and quinoline derivatives as P. falciparum inhibitors (3D7 strain). Thirty compounds exhibited anti-P. falciparum activity, with IC50 values ranging from 0.030 to 9.1 mM. The [1,2,4]triazolo[1,5-a]pyrimidine derivatives were more potent than the pyrazolo[1,5-a]pyrimidine and quinoline analogues. Compounds 20, 21, 23 and 24 were the most potent inhibitors, with IC50 values in the range of 0.030e0.086 mM and were equipotent to chloroquine. In addition, the compounds were selective, showing no cytotoxic activity against the human hepatoma cell line HepG2. All [1,2,4]triazolo[1,5-a]pyrimidine derivatives inhibited PfDHODH activity in the low micromolar to low nanomolar range (IC50 values of 0.08e1.3 mM) and did not show significant inhibition against the HsDHODH homologue (0e30% at 50 mM). Molecular docking studies indicated the binding mode of [1,2,4]triazolo[1,5-a]pyrimidine derivatives to PfDHODH, and the highest interaction affinities for the PfDHODH enzyme were in agreement with the in vitro experimental evaluation. Thus, the most active compounds against P. falciparum parasites 20 (R ¼ CF3, R1 ¼ F; IC50 ¼ 0.086 mM), 21 (R ¼ CF3; R1 ¼ CH3; IC50 ¼ 0.032 mM), 23, (R ¼ CF3, R1 ¼ CF3; IC50 ¼ 0.030 mM) and 24 (R ¼ CF3, 2- naphthyl; IC50 ¼ 0.050 mM) and the most active inhibitor against PfDHODH 19 (R ¼ CF3, R1 ¼ Cl; IC50 ¼ 0.08 mM - PfDHODH) stood out as new lead compounds for antimalarial drug discovery. Their potent in vitro activity against P. falciparum and the selective inhibition of the PfDHODH enzyme strongly suggest that this is the mechanism of action underlying this series of new [1,2,4]triazolo[1,5-a]pyrimidine derivatives. Abstract: In this work, we designed and synthesized 35 new triazolopyrimidine, pyrazolopyrimidine and quinoline derivatives as P. falciparum inhibitors (3D7 strain). Thirty compounds exhibited anti-P. falciparum activity, with IC50 values ranging from 0.030 to 9.1 mM. The [1,2,4]triazolo[1,5-a]pyrimidine derivatives were more potent than the pyrazolo[1,5-a]pyrimidine and quinoline analogues. Compounds 20, 21, 23 and 24 were the most potent inhibitors, with IC50 values in the range of 0.030e0.086 mM and were equipotent to chloroquine. In addition, the compounds were selective, showing no cytotoxic activity against the human hepatoma cell line HepG2. All [1,2,4]triazolo[1,5-a]pyrimidine derivatives inhibited PfDHODH activity in the low micromolar to low nanomolar range (IC50 values of 0.08e1.3 mM) and did not show significant inhibition against the HsDHODH homologue (0e30% at 50 mM). Molecular docking studies indicated the binding mode of [1,2,4]triazolo[1,5-a]pyrimidine derivatives to PfDHODH, and the highest interaction affinities for the PfDHODH enzyme were in agreement with the in vitro experimental evaluation. Thus, the most active compounds against P. falciparum parasites 20 (R ¼ CF3, R1 ¼ F; IC50 ¼ 0.086 mM), 21 (R ¼ CF3; R1 ¼ CH3; IC50 ¼ 0.032 mM), 23, (R ¼ CF3, R1 ¼ CF3; IC50 ¼ 0.030 mM) and 24 (R ¼ CF3, 2- naphthyl; IC50 ¼ 0.050 mM) and the most active inhibitor against PfDHODH 19 (R ¼ CF3, R1 ¼ Cl; IC50 ¼ 0.08 mM - PfDHODH) stood out as new lead compounds for antimalarial drug discovery. Their potent in vitro activity against P. falciparum and the selective inhibition of the PfDHODH enzyme strongly suggest that this is the mechanism of action underlying this series of new [1,2,4]triazolo[1,5-a]pyrimidine derivatives. |
| European Journal of Medicinal Chemistry |
| v. 209, p. 112941-1-112941-16 - Ano: 2021 |
| Fator de Impacto: 5,572 |
| |
 @article={003013328,author = {SILVEIRA, Flávia F.; SOUZA, Juliana Oliveira de; HOELZ, Lucas V. B.; CAMPOS, Vinícius R.; JABOR, Valquíria Aparecida Polisel; AGUIAR, Anna Caroline Campos; NONATO, Maria Cristina; ALBUQUERQUE, Magaly G.; GUIDO, Rafael Victorio Carvalho; BOECHAT, Nubia; PINHEIRO, Luiz C. S.},title={Comparative study between the anti-P. falciparum activity of triazolopyrimidine, pyrazolopyrimidine and quinoline derivatives and the identification of new PfDHODH inhibitors},journal={European Journal of Medicinal Chemistry},note={v. 209, p. 112941-1-112941-16},year={2021}} @article={003013328,author = {SILVEIRA, Flávia F.; SOUZA, Juliana Oliveira de; HOELZ, Lucas V. B.; CAMPOS, Vinícius R.; JABOR, Valquíria Aparecida Polisel; AGUIAR, Anna Caroline Campos; NONATO, Maria Cristina; ALBUQUERQUE, Magaly G.; GUIDO, Rafael Victorio Carvalho; BOECHAT, Nubia; PINHEIRO, Luiz C. S.},title={Comparative study between the anti-P. falciparum activity of triazolopyrimidine, pyrazolopyrimidine and quinoline derivatives and the identification of new PfDHODH inhibitors},journal={European Journal of Medicinal Chemistry},note={v. 209, p. 112941-1-112941-16},year={2021}} |





 Abstract: In this work, we designed and synthesized 35 new triazolopyrimidine, pyrazolopyrimidine and quinoline derivatives as P. falciparum inhibitors (3D7 strain). Thirty compounds exhibited anti-P. falciparum activity, with IC50 values ranging from 0.030 to 9.1 mM. The [1,2,4]triazolo[1,5-a]pyrimidine derivatives were more potent than the pyrazolo[1,5-a]pyrimidine and quinoline analogues. Compounds 20, 21, 23 and 24 were the most potent inhibitors, with IC50 values in the range of 0.030e0.086 mM and were equipotent to chloroquine. In addition, the compounds were selective, showing no cytotoxic activity against the human hepatoma cell line HepG2. All [1,2,4]triazolo[1,5-a]pyrimidine derivatives inhibited PfDHODH activity in the low micromolar to low nanomolar range (IC50 values of 0.08e1.3 mM) and did not show significant inhibition against the HsDHODH homologue (0e30% at 50 mM). Molecular docking studies indicated the binding mode of [1,2,4]triazolo[1,5-a]pyrimidine derivatives to PfDHODH, and the highest interaction affinities for the PfDHODH enzyme were in agreement with the in vitro experimental evaluation. Thus, the most active compounds against P. falciparum parasites 20 (R ¼ CF3, R1 ¼ F; IC50 ¼ 0.086 mM), 21 (R ¼ CF3; R1 ¼ CH3; IC50 ¼ 0.032 mM), 23, (R ¼ CF3, R1 ¼ CF3; IC50 ¼ 0.030 mM) and 24 (R ¼ CF3, 2- naphthyl; IC50 ¼ 0.050 mM) and the most active inhibitor against PfDHODH 19 (R ¼ CF3, R1 ¼ Cl; IC50 ¼ 0.08 mM - PfDHODH) stood out as new lead compounds for antimalarial drug discovery. Their potent in vitro activity against P. falciparum and the selective inhibition of the PfDHODH enzyme strongly suggest that this is the mechanism of action underlying this series of new [1,2,4]triazolo[1,5-a]pyrimidine derivatives.
Abstract: In this work, we designed and synthesized 35 new triazolopyrimidine, pyrazolopyrimidine and quinoline derivatives as P. falciparum inhibitors (3D7 strain). Thirty compounds exhibited anti-P. falciparum activity, with IC50 values ranging from 0.030 to 9.1 mM. The [1,2,4]triazolo[1,5-a]pyrimidine derivatives were more potent than the pyrazolo[1,5-a]pyrimidine and quinoline analogues. Compounds 20, 21, 23 and 24 were the most potent inhibitors, with IC50 values in the range of 0.030e0.086 mM and were equipotent to chloroquine. In addition, the compounds were selective, showing no cytotoxic activity against the human hepatoma cell line HepG2. All [1,2,4]triazolo[1,5-a]pyrimidine derivatives inhibited PfDHODH activity in the low micromolar to low nanomolar range (IC50 values of 0.08e1.3 mM) and did not show significant inhibition against the HsDHODH homologue (0e30% at 50 mM). Molecular docking studies indicated the binding mode of [1,2,4]triazolo[1,5-a]pyrimidine derivatives to PfDHODH, and the highest interaction affinities for the PfDHODH enzyme were in agreement with the in vitro experimental evaluation. Thus, the most active compounds against P. falciparum parasites 20 (R ¼ CF3, R1 ¼ F; IC50 ¼ 0.086 mM), 21 (R ¼ CF3; R1 ¼ CH3; IC50 ¼ 0.032 mM), 23, (R ¼ CF3, R1 ¼ CF3; IC50 ¼ 0.030 mM) and 24 (R ¼ CF3, 2- naphthyl; IC50 ¼ 0.050 mM) and the most active inhibitor against PfDHODH 19 (R ¼ CF3, R1 ¼ Cl; IC50 ¼ 0.08 mM - PfDHODH) stood out as new lead compounds for antimalarial drug discovery. Their potent in vitro activity against P. falciparum and the selective inhibition of the PfDHODH enzyme strongly suggest that this is the mechanism of action underlying this series of new [1,2,4]triazolo[1,5-a]pyrimidine derivatives. @article={003013328,author = {SILVEIRA, Flávia F.; SOUZA, Juliana Oliveira de; HOELZ, Lucas V. B.; CAMPOS, Vinícius R.; JABOR, Valquíria Aparecida Polisel; AGUIAR, Anna Caroline Campos; NONATO, Maria Cristina; ALBUQUERQUE, Magaly G.; GUIDO, Rafael Victorio Carvalho; BOECHAT, Nubia; PINHEIRO, Luiz C. S.},title={Comparative study between the anti-P. falciparum activity of triazolopyrimidine, pyrazolopyrimidine and quinoline derivatives and the identification of new PfDHODH inhibitors},journal={European Journal of Medicinal Chemistry},note={v. 209, p. 112941-1-112941-16},year={2021}}
@article={003013328,author = {SILVEIRA, Flávia F.; SOUZA, Juliana Oliveira de; HOELZ, Lucas V. B.; CAMPOS, Vinícius R.; JABOR, Valquíria Aparecida Polisel; AGUIAR, Anna Caroline Campos; NONATO, Maria Cristina; ALBUQUERQUE, Magaly G.; GUIDO, Rafael Victorio Carvalho; BOECHAT, Nubia; PINHEIRO, Luiz C. S.},title={Comparative study between the anti-P. falciparum activity of triazolopyrimidine, pyrazolopyrimidine and quinoline derivatives and the identification of new PfDHODH inhibitors},journal={European Journal of Medicinal Chemistry},note={v. 209, p. 112941-1-112941-16},year={2021}}