Low-resolution molecular shape, biochemical characterization and emulsification properties of a halotolerant esterase from Bacillus licheniformis.
LEITE, Ana Elisa Tognoli; BRIGANTI, Lorenzo Eugene Robert Pietro Paolo Lodovico Dias Blattlin; ARAÚJO, Evandro Ares de; PELLEGRINI, Vanessa de Oliveira Arnoldi Cesar Moyses Camilo1; CAMILO, Cesar Moyses; POLIKARPOV, Igor.
LEITE, Ana Elisa Tognoli; BRIGANTI, Lorenzo Eugene Robert Pietro Paolo Lodovico Dias Blattlin; ARAÚJO, Evandro Ares de; PELLEGRINI, Vanessa de Oliveira Arnoldi Cesar Moyses Camilo1; CAMILO, Cesar Moyses; POLIKARPOV, Igor.





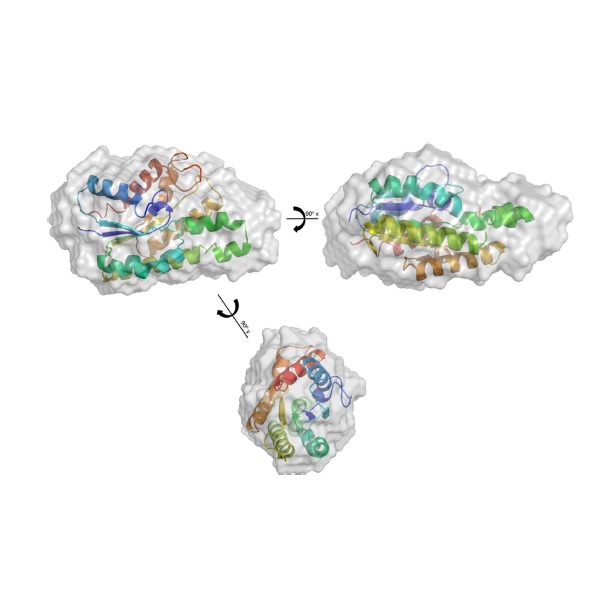 Abstract: Bacterial esterases are highly versatile enzymes, currently widely used in detergents, biosurfactants, bioemulsifiers and as biocatalysts in paper and food industries. Present work describes heterologous expression, purification, and biophysical and biochemical characterization of a halotolerant esterase from Bacillus licheniformis (BlEstA). BlEstA preferentially cleaves pNP-octanoate and both activity and stability of the enzyme increased in the presence of 2 M NaCl, and also with several organic solvents (ethanol, methanol and DMSO). Furthermore, BlEstA has considerable emulsifying properties, particularly with olive oil as substrate. Our studies also show that the enzyme is monomeric in solution and its small-angle X-ray scattering low-resolution molecular envelope fits well its high-resolution homology model.
Abstract: Bacterial esterases are highly versatile enzymes, currently widely used in detergents, biosurfactants, bioemulsifiers and as biocatalysts in paper and food industries. Present work describes heterologous expression, purification, and biophysical and biochemical characterization of a halotolerant esterase from Bacillus licheniformis (BlEstA). BlEstA preferentially cleaves pNP-octanoate and both activity and stability of the enzyme increased in the presence of 2 M NaCl, and also with several organic solvents (ethanol, methanol and DMSO). Furthermore, BlEstA has considerable emulsifying properties, particularly with olive oil as substrate. Our studies also show that the enzyme is monomeric in solution and its small-angle X-ray scattering low-resolution molecular envelope fits well its high-resolution homology model. @article={003004161,author = {LEITE, Ana Elisa Tognoli; BRIGANTI, Lorenzo Eugene Robert Pietro Paolo Lodovico Dias Blattlin; ARAÚJO, Evandro Ares de; PELLEGRINI, Vanessa de Oliveira Arnoldi Cesar Moyses Camilo1; CAMILO, Cesar Moyses; POLIKARPOV, Igor.},title={Low-resolution molecular shape, biochemical characterization and emulsification properties of a halotolerant esterase from Bacillus licheniformis},journal={European Biophysics Journal},note={v. 49, n. 6, p. 435-447},year={2020}}
@article={003004161,author = {LEITE, Ana Elisa Tognoli; BRIGANTI, Lorenzo Eugene Robert Pietro Paolo Lodovico Dias Blattlin; ARAÚJO, Evandro Ares de; PELLEGRINI, Vanessa de Oliveira Arnoldi Cesar Moyses Camilo1; CAMILO, Cesar Moyses; POLIKARPOV, Igor.},title={Low-resolution molecular shape, biochemical characterization and emulsification properties of a halotolerant esterase from Bacillus licheniformis},journal={European Biophysics Journal},note={v. 49, n. 6, p. 435-447},year={2020}}