Structural insights into the hydrolysis pattern and molecular dynamics simulations of GH45 subfamily a endoglucanase from Neurospora crassa OR74A.
KADOWAKI, Marco Antonio Seiki; POLIKARPOV, Igor.
KADOWAKI, Marco Antonio Seiki; POLIKARPOV, Igor.
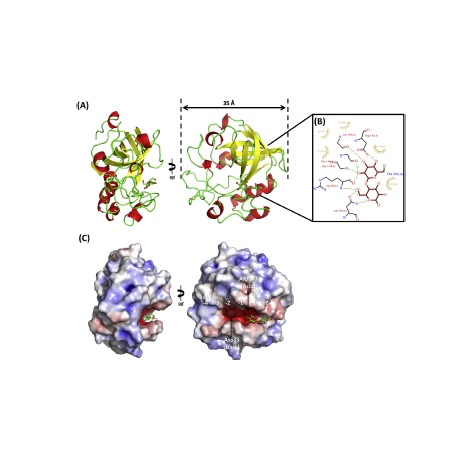 Abstract: Glycoside hydrolase (GH) family 45 is one of the smallest and poorly studied endoglucanase family with a broad biotechnological application ranging from treatment of textiles to conversion of complex cell wall polysaccharides into simple oligo- and monosaccharides. In a present study, GH45 cellulase from Neurospora crassa OR74A (NcCel45A) was characterized both biochemically and structurally. HPLC analysis of the hydrolytic products confirmed the endo-b(1,4) mode of action of the enzyme. Moreover, such pattern revealed that NcCel45A cannot hydrolyze efficiently oligosaccharides with a degree of polymerization smaller than six. The crystal structure of NcCel45A catalytic domain in the apo-form was determined at 1.9 Å resolution and the structure of the enzyme bound to cellobiose was solved and refined to 1.8 Å resolution. Comparative structural analyses and molecular dynamics simulations show that the enzyme dynamics is affected by substrate binding. Taken together, MD simulations and statistical coupling analysis revealed previously unknown correlation of a loop 6 with the breakdown of cellulose substrates by GH45. Abstract: Glycoside hydrolase (GH) family 45 is one of the smallest and poorly studied endoglucanase family with a broad biotechnological application ranging from treatment of textiles to conversion of complex cell wall polysaccharides into simple oligo- and monosaccharides. In a present study, GH45 cellulase from Neurospora crassa OR74A (NcCel45A) was characterized both biochemically and structurally. HPLC analysis of the hydrolytic products confirmed the endo-b(1,4) mode of action of the enzyme. Moreover, such pattern revealed that NcCel45A cannot hydrolyze efficiently oligosaccharides with a degree of polymerization smaller than six. The crystal structure of NcCel45A catalytic domain in the apo-form was determined at 1.9 Å resolution and the structure of the enzyme bound to cellobiose was solved and refined to 1.8 Å resolution. Comparative structural analyses and molecular dynamics simulations show that the enzyme dynamics is affected by substrate binding. Taken together, MD simulations and statistical coupling analysis revealed previously unknown correlation of a loop 6 with the breakdown of cellulose substrates by GH45. | |
| Biochimie |
| v. 165, p. 275-284 - Ano: 2019 |
| Fator de Impacto: 3,362 |
| http://dx.doi.org/10.1016/j.biochi.2019.08.016 |  @article={002966506,author = {KADOWAKI, Marco Antonio Seiki; POLIKARPOV, Igor.},title={Structural insights into the hydrolysis pattern and molecular dynamics simulations of GH45 subfamily a endoglucanase from Neurospora crassa OR74A},journal={Biochimie},note={v. 165, p. 275-284},year={2019}} @article={002966506,author = {KADOWAKI, Marco Antonio Seiki; POLIKARPOV, Igor.},title={Structural insights into the hydrolysis pattern and molecular dynamics simulations of GH45 subfamily a endoglucanase from Neurospora crassa OR74A},journal={Biochimie},note={v. 165, p. 275-284},year={2019}} |




