Unveiling the photophysical, biomolecule binding and photo-oxidative capacity of novel Ru(II)-polypyridyl corroles: a multipronged approach.
ACUNHA, Thiago V.; RODRIGUES, Bruna M.; SILVA, Júlia Akiyama da; GALINDO, Danyellen D. M.; CHAVES, Otávio A.; ROCHA, Vinícius N. da; PIQUINI, Paulo C.; KÖHLER, Mateus H.; DE BONI, Leonardo; IGLESIAS, Bernardo A.
ACUNHA, Thiago V.; RODRIGUES, Bruna M.; SILVA, Júlia Akiyama da; GALINDO, Danyellen D. M.; CHAVES, Otávio A.; ROCHA, Vinícius N. da; PIQUINI, Paulo C.; KÖHLER, Mateus H.; DE BONI, Leonardo; IGLESIAS, Bernardo A.




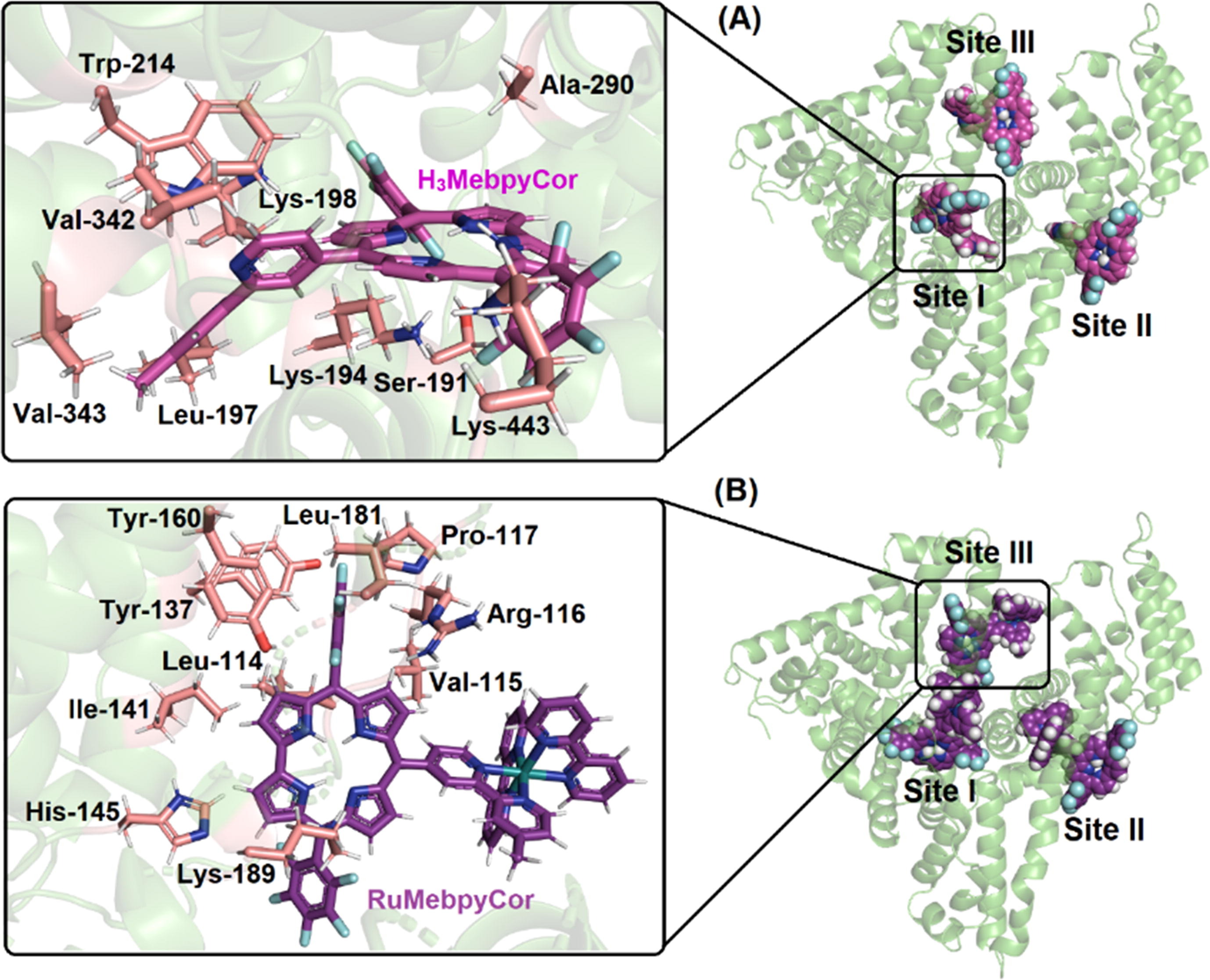 Abstract: Two fluorinated corrole derivatives, namely H3MebpyCor and RuMebpyCor, were synthesized and characterized in terms of their photophysical, electrochemical, and photooxidation behavior. Their biomolecular binding capacity toward Human Serum Albumin (HSA) and calf thymus DNA (CT-DNA) was also evaluated. The experimental data were correlated with theoretical analysis via Time Dependent Density Functional Theory (TDDFT) and Molecular Docking calculations. We found an extremely low fluorescence quantum yield for both corroles dissolved in acetonitrile (CH3CN) and dimethyl sulfoxide (DMSO), which suggests a relaxation pathway through nonradiative processes, such as internal conversion. However, the internal conversion channel in RuMebpyCor competes with the energy transfer to the Ru(II) moiety, placing triplet state formation as a second mechanism of relaxation for this structure. Both corroles showed no aggregation and good photostability. The singlet oxygen (1O2) quantum yield (F?) values also highlight their potential application in photoinactivation approaches. Theoretical analysis suggests that the introduction of Ru(II) ion coordinated in the bipyridyl unit significantly alters the optically induced electronic transitions, and that the solvents play an important role in defining the optical spectra of the two corroles. Additionally, they interact spontaneously and moderate to strong via ground-state association (static process) with HSA and CT-DNA. However, for the HSA binding assays the dynamic fluorescence quenching mechanism can also occur at high corrole concentrations. Finally, in silico calculations suggest that the main binding region into CT-DNA is the minor groove, while subdomain IIA (site I) is the main binding region for HSA:H3MebpyCor (experimental proportion 1:1) and both subdomains IIA and IB (sites I and III, respectively) for HSA:RuMebpyCor (experimental proportion 1:2).
Abstract: Two fluorinated corrole derivatives, namely H3MebpyCor and RuMebpyCor, were synthesized and characterized in terms of their photophysical, electrochemical, and photooxidation behavior. Their biomolecular binding capacity toward Human Serum Albumin (HSA) and calf thymus DNA (CT-DNA) was also evaluated. The experimental data were correlated with theoretical analysis via Time Dependent Density Functional Theory (TDDFT) and Molecular Docking calculations. We found an extremely low fluorescence quantum yield for both corroles dissolved in acetonitrile (CH3CN) and dimethyl sulfoxide (DMSO), which suggests a relaxation pathway through nonradiative processes, such as internal conversion. However, the internal conversion channel in RuMebpyCor competes with the energy transfer to the Ru(II) moiety, placing triplet state formation as a second mechanism of relaxation for this structure. Both corroles showed no aggregation and good photostability. The singlet oxygen (1O2) quantum yield (F?) values also highlight their potential application in photoinactivation approaches. Theoretical analysis suggests that the introduction of Ru(II) ion coordinated in the bipyridyl unit significantly alters the optically induced electronic transitions, and that the solvents play an important role in defining the optical spectra of the two corroles. Additionally, they interact spontaneously and moderate to strong via ground-state association (static process) with HSA and CT-DNA. However, for the HSA binding assays the dynamic fluorescence quenching mechanism can also occur at high corrole concentrations. Finally, in silico calculations suggest that the main binding region into CT-DNA is the minor groove, while subdomain IIA (site I) is the main binding region for HSA:H3MebpyCor (experimental proportion 1:1) and both subdomains IIA and IB (sites I and III, respectively) for HSA:RuMebpyCor (experimental proportion 1:2). @article={003039897,author = {ACUNHA, Thiago V.; RODRIGUES, Bruna M.; SILVA, Júlia Akiyama da; GALINDO, Danyellen D. M.; CHAVES, Otávio A.; ROCHA, Vinícius N. da; PIQUINI, Paulo C.; KÖHLER, Mateus H.; DE BONI, Leonardo; IGLESIAS, Bernardo A.},title={Unveiling the photophysical, biomolecule binding and photo-oxidative capacity of novel Ru(II)-polypyridyl corroles: a multipronged approach},journal={Journal of Molecular Liquids},note={v. 340, p. 117223-1-117223-16 + supplementary material},year={2021}}
@article={003039897,author = {ACUNHA, Thiago V.; RODRIGUES, Bruna M.; SILVA, Júlia Akiyama da; GALINDO, Danyellen D. M.; CHAVES, Otávio A.; ROCHA, Vinícius N. da; PIQUINI, Paulo C.; KÖHLER, Mateus H.; DE BONI, Leonardo; IGLESIAS, Bernardo A.},title={Unveiling the photophysical, biomolecule binding and photo-oxidative capacity of novel Ru(II)-polypyridyl corroles: a multipronged approach},journal={Journal of Molecular Liquids},note={v. 340, p. 117223-1-117223-16 + supplementary material},year={2021}}